Table of Contents
- What Is EU REACH?
- A Brief History of REACH
- Key Lists of the REACH regulation framework
- Five-Step Guide to REACH Compliance
- REACH Compliance Workflow
- REACH Compliance: Article vs. Substance Manufacturers
- Ongoing REACH Compliance Obligations
- REACH in Other Jurisdictions
- Still Managing REACH Manually?
- Your Next Move
What Is EU REACH?
REACH isn’t optional. If you make, import, or sell products in the EU that contain chemicals—even inside finished goods—you’re on the hook. Get it right, or risk losing market access.
REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) is the EU regulation that governs chemical safety. It puts the burden of proof on businesses. If you can’t prove safety, you can’t sell.
You must:
- Register substances with ECHA if usage exceeds 1 tonne/year.
- Evaluate chemical safety and manage risks.
- Notify customers and authorities of SVHCs.
- Apply for authorization or follow use restrictions.
Related: The Complete Guide to EU REACH Regulation
A Brief History of REACH
- Adopted: December 2006
- In Force: June 2007 REACH replaced fragmented chemical laws with a single, directly applicable regulation across the EU. It’s governed by the European Chemicals Agency (ECHA).
Related: REACH Candidate List Update: Jan 2025 SVHC Additions
Key Lists of the REACH regulation framework
- Annex XVII Guide: Restricted Chemicals
- Annex XIV Explained: Authorisation Pathways
- Candidate List: Definition: The Candidate List is a compilation of substances that ECHA is currently evaluating due to their potential adverse effects on human health or the environment.
Purpose: These substances are under consideration for inclusion in the Authorization List. Firms using substances from the Candidate List are encouraged to explore alternative, safer options.
- Authorisation List: Definition: The Authorisation List (Annex XIV) consists of substances that have been identified as having serious effects on health or the environment.
Purpose: Substances listed in Annex XIV require explicit authorization for their use. Companies must apply for authorization to continue using these substances, and there is a sunset date after which use without authorization is prohibited.
- Restricted List: Definition: The Restricted List (Annex XVII) comprises substances that ECHA has either banned or restricted in specific situations or for certain uses.
Purpose: Restrictions may include limitations on concentration levels or usage in specific applications to mitigate risks to human health and the environment. Compliance with the restrictions outlined in Annex XVII is mandatory.

Five-Step Guide to REACH Compliance
- Collect Substance Information Dig deep into your supply chain. You need full material disclosures, SDSs, and any test reports—especially for substances >1 tonne/year.
- Register with ECHA Submit a joint registration dossier under the “one substance, one registration” rule. No shortcut here.
- ECHA Evaluation They’ll review your data and might ask for more. Don’t get caught with weak documentation.
- Authorization & Restriction Rulings If your substances are risky, you’ll need to prove safe use or stop using them altogether.
- Get Your REACH Registration Number No number, no compliance. That’s your regulatory receipt. Keep it audit-ready.

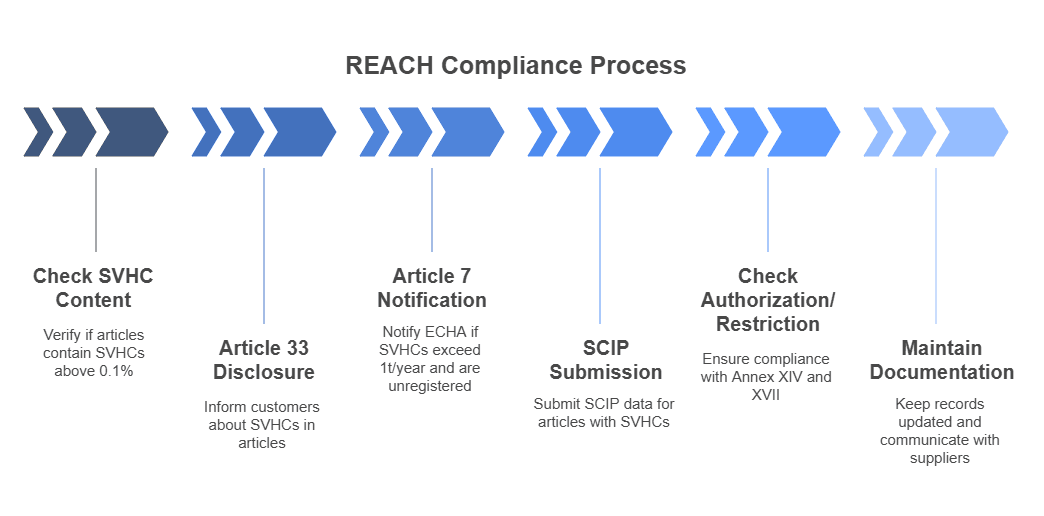
REACH Compliance Workflow
- Check SVHC content (>0.1%) in articles
- Article 33 Disclosure to customers and upon request
- Article 7 Notification to ECHA (if >1t/year and not registered)
- SCIP Submission for all articles with SVHCs (since 2021)
- Check for Authorization (Annex XIV) or Restriction (Annex XVII)
- Maintain updated documentation and supplier outreach
Related: SCIP Database vs. REACH Reporting Requirements
REACH Compliance: Article vs. Substance Manufacturers
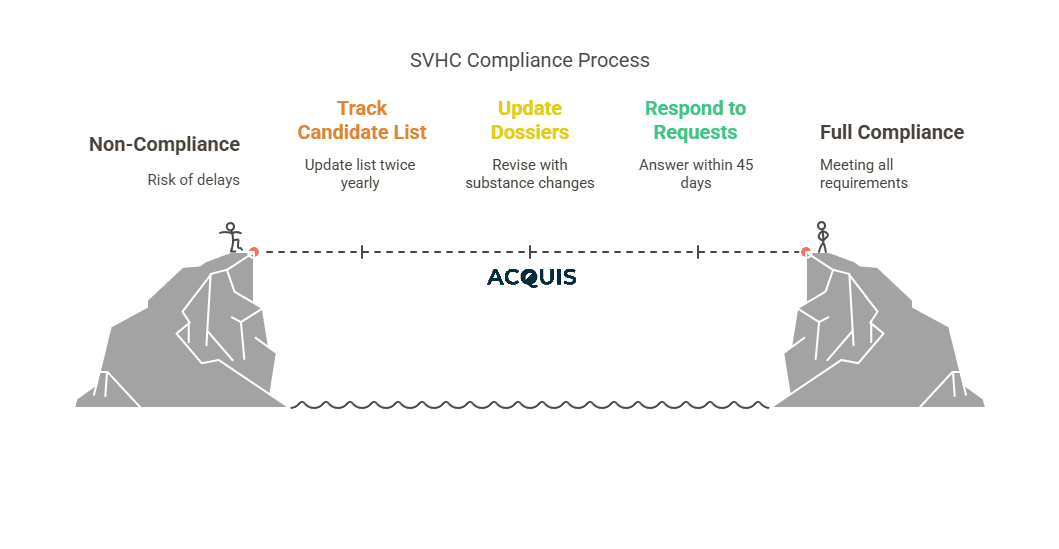
Ongoing REACH Compliance Obligations
- Track the Candidate List (updated twice yearly).
- Update dossiers if substance volume, use, or hazards change.
- Respond to SVHC requests under Article 33 within 45 days—no delays.
REACH in Other Jurisdictions
- UK REACH: Post-Brexit version of REACH
- Ukraine REACH: REACH-aligned regulation (Resolution 847)
Still Managing REACH Manually?
You're burning time, budget, and audit readiness.
Acquis Compliance Platform does the heavy lifting:
- Automates supplier requests and validation
- Flags SVHCs in real time across BOMs
- Prepares SCIP dossiers in minutes
- Tracks changes in EU regulations by substance and market
With Acquis, you get:
- Centralized documentation
- 80%+ manual effort reduction
- Total traceability across every part and supplier
Your Next Move
If your REACH program still runs on spreadsheets, it’s not compliant—it’s at risk.
Get ahead of the next SVHC update. Book a demo with Acquis today.
